An Introduction
People with liver pathology have been shown to have a higher amount of circulating LPS due to reduced liver functioning and increased intestinal permeability (1, 2). Moreover, the degree of endotoxemia is correlated with the degree of liver failure (3).
LPS, also referred to as endotoxins, are large molecules that consist of a lipid and polysaccharide joined together. They are an important constituent of gram-negative bacteria and cyanobacteria cell walls and are released in large numbers when the cell is broken down or the colony grows in large amounts (4).
Gram-negative bacteria are commensal inhabitants in our microbiome and in small amounts they play an important role in building our innate immunity (5). However, LPS are able to initiate a vigorous inflammatory response. Indeed, they are so effective at inducing inflammation in the body, that they are often the basis of creating inflammatory models in research trials (6). In high quantities LPS in the blood can lead to septicaemia, but exposure to high levels of LPS is also indicated as a driving factor in many other inflammatory disorders, such as metabolic syndrome, liver and renal diseases, chronic obstructive pulmonary disease, cardiovascular disease, autoimmunity and neurodegenerative disorders, amongst others (4,7-10). In order to tightly regulate our exposure to them, we have developed a strong immune response. Central to this, is the liver.
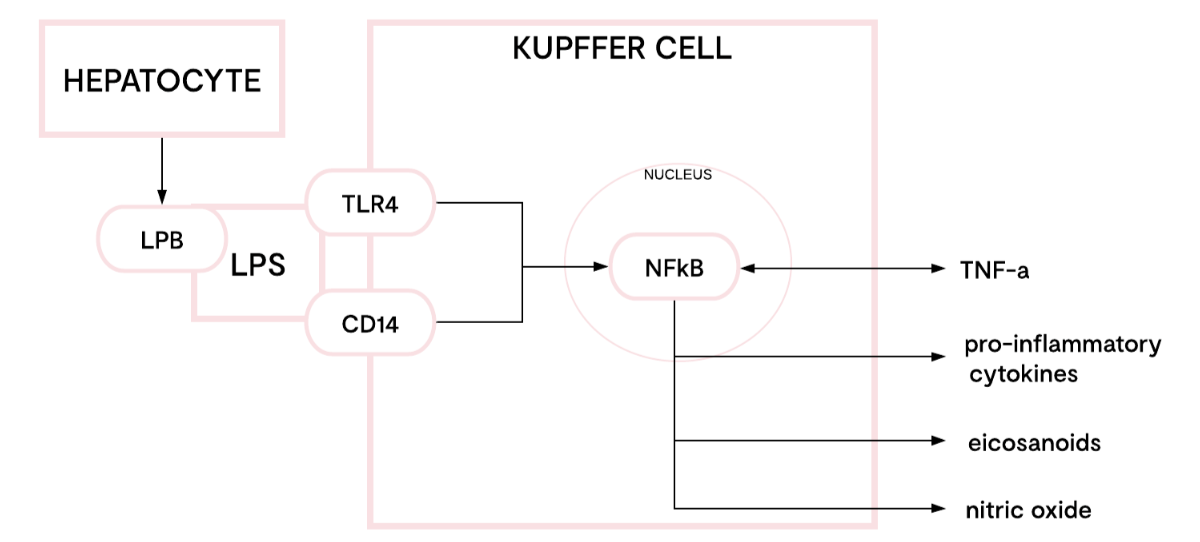
Fig 1. One pathway of lipopolysaccharide (LPS) signalling in Kupffer cells.
Adapted from Su, G. (12).
The liver is the final barrier to prevent GI bacteria and bacterial products such as LPS and other toxins from entering the systemic blood stream (11). Bile is part of this strategy as it is a potent antimicrobial agent (12). The liver is also the primary site of excretion, since LPS are treated as lipoproteins in the body.
In addition to LPS clearance, the liver is a key site of LPS sampling and immune initiation. Kupffer cells reside in the liver sinusoids, which drain blood from the GI tract, therefore they are chronically exposed to LPS. LPS activate Kupffer cells primarily by three mechanisms: increasing expression of CD14, TLR-4 binding, and binding to LPS binding protein (LPB) which is produced by hepatocytes (See Fig 1). The resultant initiation of cytokines, including IL-8 and TNF-a, eicaosanoids, nitric oxide, and reactive oxygen intermediates via NF-kB translocation, aids the immune response to, and clearance of, LPS (13).




